| Bivalve Molluscs |
Bivalve molluscs are so-called because their shell is divided into two articulated valves (compare them to gastropods
and cephalopods, which are also molluscs). In large bivalves, such as scallops and large clams, the valves are
corrugated to give them extra mechanical strength. Most bivalves are not capable of active swimming and most are
burrowing animals, using their muscular foot to excavate a burrow in soft sediments (some bore into corals and similar
hard materials). The animal typically sits with its anterior end uppermost and agape to allow water to enter for
respiratory purposes (driven by ciliary action). Many have also adapted the respiratory currents and modified their
gills to also filter out organic particles from the inhalant water. Cilia then convey these particles to the mouth. The
detailed anatomy of a scallop is shown below (showing the animal sitting in the right valve).
and cephalopods, which are also molluscs). In large bivalves, such as scallops and large clams, the valves are
corrugated to give them extra mechanical strength. Most bivalves are not capable of active swimming and most are
burrowing animals, using their muscular foot to excavate a burrow in soft sediments (some bore into corals and similar
hard materials). The animal typically sits with its anterior end uppermost and agape to allow water to enter for
respiratory purposes (driven by ciliary action). Many have also adapted the respiratory currents and modified their
gills to also filter out organic particles from the inhalant water. Cilia then convey these particles to the mouth. The
detailed anatomy of a scallop is shown below (showing the animal sitting in the right valve).
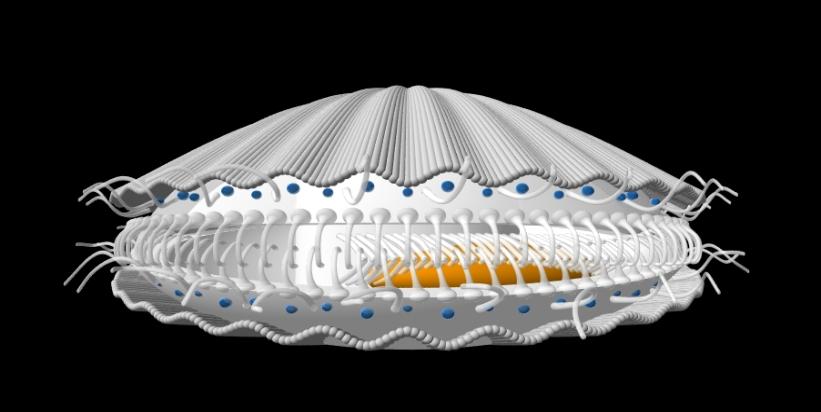
Above: a model of a scallop. Scallops are surface-dwelling bivalve molluscs which can swim by rapidly clapping
the two halves of their shells (valves) together repeatedly creating a pulsatile jet of water (a form of jet
propulsion). Each valve is lined by a lobe of tissue, called a mantle lobe. Note the bright blue eyes arrayed
around the rim of each mantle lobe, along with guard tentacles and longer sensory tentacles. Details of the
shell are shown below (a 3D computer model generate din Pov-Ray using a sinusoidal sweep of sphere-sweeps).
Note that the valves cover the left and right halves of the animal, which is laterally compressed. The anterior end
is on the left in the image above. Scallops thus swim and rest on their sides, with the deeper upper valve
representing the right-side (right valve) and the shallower bottom valve the left side (left valve). Generally,
swimming is an escape response, e.g. when touched by a predatory starfish, and the scallop swims a meter or
so, before sinking back to the bottom. It may expel a jet of water to excavate a pit in the bottom sediment in which
it rests. However, some longer migratory movements have been reported.
the two halves of their shells (valves) together repeatedly creating a pulsatile jet of water (a form of jet
propulsion). Each valve is lined by a lobe of tissue, called a mantle lobe. Note the bright blue eyes arrayed
around the rim of each mantle lobe, along with guard tentacles and longer sensory tentacles. Details of the
shell are shown below (a 3D computer model generate din Pov-Ray using a sinusoidal sweep of sphere-sweeps).
Note that the valves cover the left and right halves of the animal, which is laterally compressed. The anterior end
is on the left in the image above. Scallops thus swim and rest on their sides, with the deeper upper valve
representing the right-side (right valve) and the shallower bottom valve the left side (left valve). Generally,
swimming is an escape response, e.g. when touched by a predatory starfish, and the scallop swims a meter or
so, before sinking back to the bottom. It may expel a jet of water to excavate a pit in the bottom sediment in which
it rests. However, some longer migratory movements have been reported.
Scallops have a very well developed adductor muscle. The adductor muscle connects the two valves and closes the
shell valves together when it contracts. Bivalves generally have one pair of these muscles, one anterior and one
posterior, but only the posterior adductor muscle is developed in scallops. This large muscle, along with the gonad
(ovary + testis) is what we eat when we eat scallops. This large muscle has two regions, one composed of striated
muscle, the other of smooth muscle. Striated muscle is capable of more rapid, more forceful contractions and is used
to close the shell rapidly, such as when swimming. The smooth muscle is slower but less fatigable and is able to keep
the valves closed for prolonged periods of time. The foot in scallops is not used for burrowing and is vestigial. The
head in bivalves is much reduced, being represented externally by two pairs of flaps or labial palps, one pair
associated with one of the pair of labial tentacles.
shell valves together when it contracts. Bivalves generally have one pair of these muscles, one anterior and one
posterior, but only the posterior adductor muscle is developed in scallops. This large muscle, along with the gonad
(ovary + testis) is what we eat when we eat scallops. This large muscle has two regions, one composed of striated
muscle, the other of smooth muscle. Striated muscle is capable of more rapid, more forceful contractions and is used
to close the shell rapidly, such as when swimming. The smooth muscle is slower but less fatigable and is able to keep
the valves closed for prolonged periods of time. The foot in scallops is not used for burrowing and is vestigial. The
head in bivalves is much reduced, being represented externally by two pairs of flaps or labial palps, one pair
associated with one of the pair of labial tentacles.
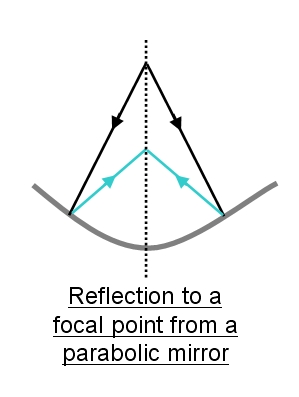
The bright blue eyes of scallops display a remarkable adaptation
which exploits physical laws in a very different way to the
mammalian eye. Instead of relying on a lens to focus the light, they
use parabolic mirrors or reflected to focus the reflected light in
much the same way as a reflector telescope used in astronomy.
This is illustrated on the left which shows the incident light (in black)
from the object reflecting from the parabolic mirror (blue rays) and
coming to a focus in front of the mirror. Telescopes generally use
mirrors made of highly polished metal.
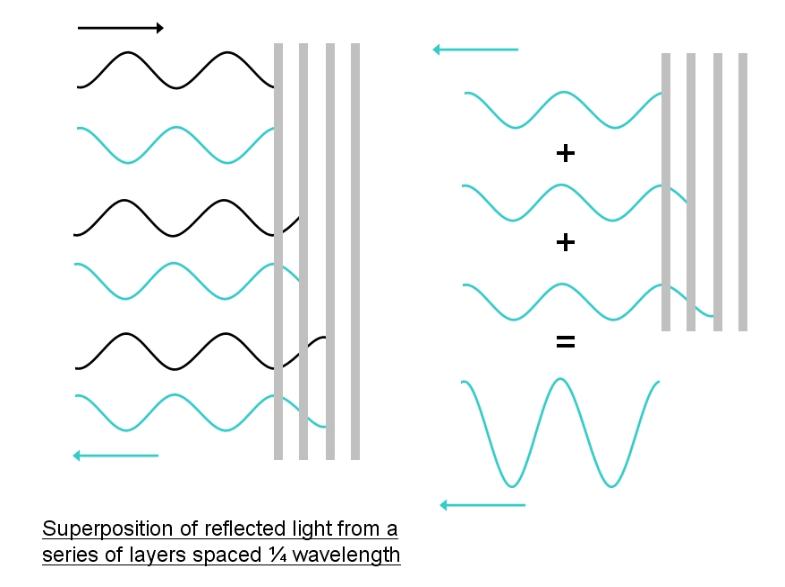
Biologically materials lack metallic lustre, but the scallop
nevertheless has reflectors as good as polished metal. This is
achieved using constructive interference. The reflective material is
arranged in several layers. The layers are spaced by a quarter of a
wavelength. Since the reflected light waves are inverted, that is
their phase is changed by 180 degrees, with a spacing of quarter
of a wavelength the reflected wave has to traverse an additional
distance of half a wavelength. This means that the light waves
reflected from each layer will be in-phase (aligned) when they
emerge from the reflector. This results in constructive interference,
which intensifies the light. Only light of the right wavelength is
reflected in this way. In the scallop eye, light of blue-green
wavelength is reflected, so the eyes look metallic blue. This
mechanism is illustrated below:
which exploits physical laws in a very different way to the
mammalian eye. Instead of relying on a lens to focus the light, they
use parabolic mirrors or reflected to focus the reflected light in
much the same way as a reflector telescope used in astronomy.
This is illustrated on the left which shows the incident light (in black)
from the object reflecting from the parabolic mirror (blue rays) and
coming to a focus in front of the mirror. Telescopes generally use
mirrors made of highly polished metal.
Biologically materials lack metallic lustre, but the scallop
nevertheless has reflectors as good as polished metal. This is
achieved using constructive interference. The reflective material is
arranged in several layers. The layers are spaced by a quarter of a
wavelength. Since the reflected light waves are inverted, that is
their phase is changed by 180 degrees, with a spacing of quarter
of a wavelength the reflected wave has to traverse an additional
distance of half a wavelength. This means that the light waves
reflected from each layer will be in-phase (aligned) when they
emerge from the reflector. This results in constructive interference,
which intensifies the light. Only light of the right wavelength is
reflected in this way. In the scallop eye, light of blue-green
wavelength is reflected, so the eyes look metallic blue. This
mechanism is illustrated below:
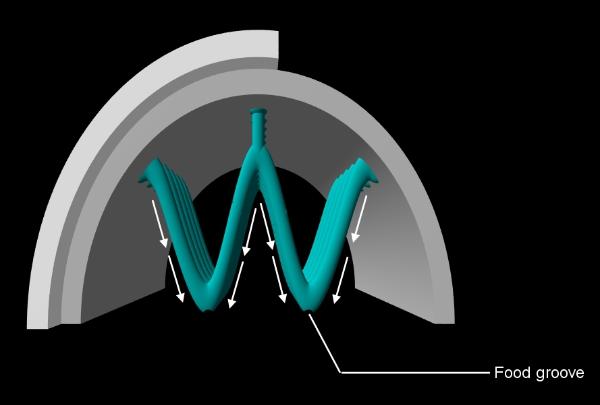
Above: in forms with two apical food grooves, the frontal cilia carry trapped particles down to apical notches.
The notches on adjacent filaments all align to form a food groove which conveys particles to the palps where
they are sorted (in much the same as the protobranch deposit feeders discussed above which use their palpal
tentacles to pick up food particles) : the particles flow across a series of ridges on the inner surface of each
palp, lighter particles continue from ridge to ridge, towards the mouth, whereas heavier particles fall into the
troughs between the ridges and are removed.
Rejected particles may fall onto the mantle floor, where further ciliary tracts carry them to the opening of the
inhalant aperture, where they accumulate as peusofaeces which are ejected when the valves periodically close,
expelling water and the pseudofaeces out through the normally inhalant opening. In non filter-feeding types
(often considered to be more archaic) the frontal cilia are used to clean the gills of sediment particles, rather
than to deliver potential food to food grooves. The gills may also carry out some sorting of particles before
these particles are further sorted by the palps. In some, heavier particles are transported away from the mouth,
to be deposited on the mantle floor and ejected as already described. Others have more elaborate sorting
mechanisms. Lamellibranchs may trap and ingest food particles as small as one micrometre in diameter (the
size of a typical bacterium).
Food particles reaching the mouth are transported to the stomach via the oesophagus, as a mucoid string. A
crystalline style (similar to that found in gastropods) is housed in the stylar sac, which connects to the
stomach. The style moves in and out of the stomach, mixing the contents and throwing them onto a ciliated
sorting 'conveyer-belt' system inside the gut. The style also gets rotated and ground against a chitinous plate,
the gastric shield, inside the stomach, by the action of cilia in the stylar sac, and hence eroded, releasing
digestive enzymes incorporated into its matrix when it is synthesised. Enzymes are also released by the stomach
wall, and digestion here is thus extracellular. New styles are regularly snthesised as needed.
Meanwhile, the sorting system of cilia conveys larger, coarser particles, which are generally inorganic and
inedible, are passed into the intestine for excretion, whereas the lighter particles are passes into the
digestive diverticula - a mass of absorptive sacs with several openings into the stomach. In the diverticula,
any utilisable food, which has already been partially digested extracellularly is phagocytosed by the cells of the
diverticula walls to be digested intracellularly and the nutrients absorbed. Indigestible material is conveyed back
into the stomach and into the intestine for excretion.
Not all bivalves feed in this manner. Some have their inhalant and exhalant apertures modified into siphons.
Fusion of the edges of the two mantle lobes together seals off the rest of the mantle cavity, such that water can
only enter through the inhalant siphon and exit through the exhalent siphon. (A third opening allows the foot
to be protruded to the exterior). These siphons can reach to the surface from deep burrows. The geoducks
(Panopea generosa) live in burrows over a meter deep and have such large siphons and bodies that neither
can be retracted into the valves. Some forms use their inhalant siphons to hoover the sea floor, drawing in
sediment particles which are sorted by the gills and palps - a form of deposit feeding.
Some deep-sea bivalves are gutless, and obtain their nutrients from symbiotic micro-organisms that live inside
them (in much the same manner as in pogonophorans). Some bivalves, the septibranchs, are predators and
scavengers. Their gills are modified to form a pair of perforated muscular septa which generate currents by
wafting up and down. The inhalant siphon is hot out towards sources of vibrations and small organisms
hoovered up! Alternatively, the siphon may have a distensible hood which closes over the prey to trap it. The
currents generated are sufficiently strong to waft small animals, such as worms and crustaceans, into the
bivalves where they are grasped by the modified (and reduced) muscular palps and passed to the mouth. They
have muscular stomachs lined by hard chitin which crush up the prey (the style is greatly reduced). Protease
enzymes secreted from the diverticula enter the stomach to begin extracellular digestion. Partially digested
particles then enter the diverticula for intracellular digestion.
From the above account one key point should be clear: the bivalves have evolved extensive and elaborate uses
for cilia. They use ciliary tracts to draw in currents of water for feeding and respiration; clean their gills; to
transport food and wastes in their guts; to sort particles in the gills, palps and inside their guts, sorting food
particles from detritus and sediment. It is hard to think of any other organism which puts cilia to such a diverse
and ingenious range of uses.
Internal Transport Systems
The bivalve heart has three chambers: two auricles (atria, input chambers) which receive blood from the gills
and a ventricle (output chamber). The heart, and usually also part of the intestine, is enclosed in the
pericardial cavity, which is the bivalve coelom (see animal bodies). The heart rate is as low as 20 bpm in a
large clam. The ventricle expels blood into the anterior aorta and sometimes also a posterior aorta. The anterior
aorta opens into tissue sinuses. Sinuses are simply tissue spaces filled with circulating blood but lacking an
endothelium or vessel wall. A circulatory system, such as this, with extensive sinuses is referred to as an open
circulatory system. Blood is circulated to the nephridia, gills and mantle before returning to the heart. The
mantle has a large surface area and contributes to gas exchange.
Some bivalves have hameoglobin, which may be extracellular (free in the plasma) or intracellular (inside red
blood cells) and which functions both to transport and to store oxygen. Myoglobin may also be present in the
muscles, acting as a further oxygen store. A few prosobranches have the blue copper-based respiratory
pigment haemocyanin instead, but most bivalves have no respiratory blood pigments.
Excretion
There are two nephridia (excretory tubules) beneath the pericardium. Each may form a U-tube (as in
Anodonta) or a long branched tube (as in Mytilus). Typically, one end of the tubule opens in the pericardial
cavity via a nephrostome (see earthworm excretion for a general discussion of invertebrate excretion) (after all
the pericardial cavity corresponds to the coelom cavity). The other end of the tubule opens into the mantle
cavity via a nephridiopore. The auricles and glands in the pericardium (pericardial glands) contain a porous
epithelium (composed of podocytes resembling podocytes in the mammalian kidney) and it is thought that
ultrafiltration occurs across these membranes to remove excess water and waste materials.
Nervous System
the nervous system is a paired structure, with pairs of ganglia and nerve cords. The 'brain' consists of a pair of
cerebropleural ganglia (fused cerebral and pleural ganglia) one on each side of the oesophagus, connected
by a dorsal commissure (connecting nerve tract). Two pairs of longitudinal nerve cords extend from these
ganglia. One pair innervates the visceral mass, terminating in visceral ganglia which innervate the posterior
adductor muscle, the siphons (if present) and the internal viscera and gills. The other pair terminate in pedal
ganglia in the foot which innervate the anterior adductor muscle and the foot. The cerebral ganglia directly
innervate the anterior adductor muscle and the palps.
The foot contains one pair of statocysts, which are near to or within the pedal ganglia but innervated by the
cerebral ganglia. These are organs of equilibrium (balance). Eyes may be present. Giant clams have thousands
of simple eyes or ocelli, which are compound eyes in ark shells. Some bivalves have eyes on the head, though
this is not typical in bivalves. The mode of operation of scallop eyes has already been discussed. The
osphradium is a patch of sensory epithelium beneath the posterior adductor muscle which is thought to
monitor the water passing through the mantle cavity.
Reproduction and Development
Most bivalves are dioecious (with separate sexes) but some are hermaphrodites (e.g. some scallops). There is
a pair of gonads, but these are often more-or-less fused into a single mass. In hermaphrodite scallops the
ventral region is the ovary, the dorsal the testis. The gonads open, via simple gonoducts, either into the
nephridium, into which the gametes are shed to exit through the nephridiopores, or else the gonoducts open
directly into the mantle cavity.
In some forms the gender changes during the course of development. In the edible oyster (Ostrea edulis) the
gender periodically switches in a single individual. The gametes are shed into the surrounding water and
fertilisation is generally external, although the female may draw in spermatozoa from the surrounding water, in
the inhalent current, to fertilise the eggs in the suprabranchial cavity (region of the mantle cavity above the gills)
where the eggs may be brooded. The eggs hatch into a trochophore larva which develops into a veliger larva
with two valves. The veliger may feed planktonically or it may be nonfeeding (lechitotrophic, that is deriving
nourishment by digesting yolk reserves) before metamorphosing into a small adult. In most freshwater bivalves
development is direct, e.g. eggs are brooded in marsupial sacs between the gill lamellae in freshwater clams
and hatch into young adult clams.
In some bivalves the veliger is modified to lead a parasitic larval life on fish. One such type of modified veliger is
the glochidium which attaches to the gills, fins or body surface of a host fish via a hook on each valve and an
adhesive thread borne on the rudimentary foot. Some clams release glochidia embedded in wormlike coloured
masses. The host fish eats this mass whilst the glochidia attach to its gills. The fish serves to help disperse the
larva. The host tissues form a cyst around the glochidium and phagocytic cells in the larva's mantle ingest the
host tissues growing around it, deriving nourishment. After 10 to 30 days the larva breaks out of the cyst, sinks
to the bottom and burrows into the sediment to continue its development into adulthood. Such a parasitic life
style requires the production of large numbers of parasites, for most larva will never locate a suitable host.
Some freshwater mussels produce about 3 million glochidia.
Bivalves live up to 150 years (possibly more in some cases) though most live between 2 and 30 years. The
scallop Placopecten requires ten years to reach its maximum diameter of 15 cm. The growth rings on the valves
have already been discussed.
Ecology
Many bivalves are soft-bottom dwellers, burrowing in the sediment. Others live attached to solid surfaces either
via byssal threads (which form the anchor or byssus) or by the cementing of one valve to the substrate. Byssal
threads are very strong protein fibres secreted by the byssus gland with the help of the foot (the thread lies in a
grrove on the pedal foot during secretion and pedal glands contribute to its formation). After being secreted and
anchored, the foot withdrawals, leaving the byssal threads connected to the byssal gland via a strong stem. the
byssal fibres tan and harden in the air. The bivalve can pull on its anchoring byssus with the byssus retractor
muscle.
Some bivalves bore into rock or coral. The giant clam Tridacna gigas (which can reach 1.37 m in diameter)
bores into coral. Blood sinuses in its mantle contain photosynthetic zooxanthellae, which utilise sunlight to
produce food for themselves and the host clam via photosynthesis. The tissues are coloured blue, green, red,
violet or brown by pigments which may act as screening pigments to shield the zooxanthellae from excess
sunlight. Boring bivalves dwell in a permanent burrow, attached to one side via byssal threads or by a sucker on
the foot. Drilling is mechanical, the anterior ends of the valves slowly scraping away the substrate (they may
have serrated edges to assist in this) but can also be chemical in which secretions soften shell or coral to
facilitate mechanical drilling. In soft rocks (shale) drilling can exceed 1 cm depth a month. Shipworms drill into
wood, such as mangrove roots, timber washed out to sea and ship hulls. They may burrow up to 2 m into the
wood and eat the sawdust produced, which is digested with the help of enzymes secreted by symbiotic bacteria.
Some bivalves live as commensals with echinoderms or crustaceans. At least one form is parasitic in sea
cucumber guts.
The notches on adjacent filaments all align to form a food groove which conveys particles to the palps where
they are sorted (in much the same as the protobranch deposit feeders discussed above which use their palpal
tentacles to pick up food particles) : the particles flow across a series of ridges on the inner surface of each
palp, lighter particles continue from ridge to ridge, towards the mouth, whereas heavier particles fall into the
troughs between the ridges and are removed.
Rejected particles may fall onto the mantle floor, where further ciliary tracts carry them to the opening of the
inhalant aperture, where they accumulate as peusofaeces which are ejected when the valves periodically close,
expelling water and the pseudofaeces out through the normally inhalant opening. In non filter-feeding types
(often considered to be more archaic) the frontal cilia are used to clean the gills of sediment particles, rather
than to deliver potential food to food grooves. The gills may also carry out some sorting of particles before
these particles are further sorted by the palps. In some, heavier particles are transported away from the mouth,
to be deposited on the mantle floor and ejected as already described. Others have more elaborate sorting
mechanisms. Lamellibranchs may trap and ingest food particles as small as one micrometre in diameter (the
size of a typical bacterium).
Food particles reaching the mouth are transported to the stomach via the oesophagus, as a mucoid string. A
crystalline style (similar to that found in gastropods) is housed in the stylar sac, which connects to the
stomach. The style moves in and out of the stomach, mixing the contents and throwing them onto a ciliated
sorting 'conveyer-belt' system inside the gut. The style also gets rotated and ground against a chitinous plate,
the gastric shield, inside the stomach, by the action of cilia in the stylar sac, and hence eroded, releasing
digestive enzymes incorporated into its matrix when it is synthesised. Enzymes are also released by the stomach
wall, and digestion here is thus extracellular. New styles are regularly snthesised as needed.
Meanwhile, the sorting system of cilia conveys larger, coarser particles, which are generally inorganic and
inedible, are passed into the intestine for excretion, whereas the lighter particles are passes into the
digestive diverticula - a mass of absorptive sacs with several openings into the stomach. In the diverticula,
any utilisable food, which has already been partially digested extracellularly is phagocytosed by the cells of the
diverticula walls to be digested intracellularly and the nutrients absorbed. Indigestible material is conveyed back
into the stomach and into the intestine for excretion.
Not all bivalves feed in this manner. Some have their inhalant and exhalant apertures modified into siphons.
Fusion of the edges of the two mantle lobes together seals off the rest of the mantle cavity, such that water can
only enter through the inhalant siphon and exit through the exhalent siphon. (A third opening allows the foot
to be protruded to the exterior). These siphons can reach to the surface from deep burrows. The geoducks
(Panopea generosa) live in burrows over a meter deep and have such large siphons and bodies that neither
can be retracted into the valves. Some forms use their inhalant siphons to hoover the sea floor, drawing in
sediment particles which are sorted by the gills and palps - a form of deposit feeding.
Some deep-sea bivalves are gutless, and obtain their nutrients from symbiotic micro-organisms that live inside
them (in much the same manner as in pogonophorans). Some bivalves, the septibranchs, are predators and
scavengers. Their gills are modified to form a pair of perforated muscular septa which generate currents by
wafting up and down. The inhalant siphon is hot out towards sources of vibrations and small organisms
hoovered up! Alternatively, the siphon may have a distensible hood which closes over the prey to trap it. The
currents generated are sufficiently strong to waft small animals, such as worms and crustaceans, into the
bivalves where they are grasped by the modified (and reduced) muscular palps and passed to the mouth. They
have muscular stomachs lined by hard chitin which crush up the prey (the style is greatly reduced). Protease
enzymes secreted from the diverticula enter the stomach to begin extracellular digestion. Partially digested
particles then enter the diverticula for intracellular digestion.
From the above account one key point should be clear: the bivalves have evolved extensive and elaborate uses
for cilia. They use ciliary tracts to draw in currents of water for feeding and respiration; clean their gills; to
transport food and wastes in their guts; to sort particles in the gills, palps and inside their guts, sorting food
particles from detritus and sediment. It is hard to think of any other organism which puts cilia to such a diverse
and ingenious range of uses.
Internal Transport Systems
The bivalve heart has three chambers: two auricles (atria, input chambers) which receive blood from the gills
and a ventricle (output chamber). The heart, and usually also part of the intestine, is enclosed in the
pericardial cavity, which is the bivalve coelom (see animal bodies). The heart rate is as low as 20 bpm in a
large clam. The ventricle expels blood into the anterior aorta and sometimes also a posterior aorta. The anterior
aorta opens into tissue sinuses. Sinuses are simply tissue spaces filled with circulating blood but lacking an
endothelium or vessel wall. A circulatory system, such as this, with extensive sinuses is referred to as an open
circulatory system. Blood is circulated to the nephridia, gills and mantle before returning to the heart. The
mantle has a large surface area and contributes to gas exchange.
Some bivalves have hameoglobin, which may be extracellular (free in the plasma) or intracellular (inside red
blood cells) and which functions both to transport and to store oxygen. Myoglobin may also be present in the
muscles, acting as a further oxygen store. A few prosobranches have the blue copper-based respiratory
pigment haemocyanin instead, but most bivalves have no respiratory blood pigments.
Excretion
There are two nephridia (excretory tubules) beneath the pericardium. Each may form a U-tube (as in
Anodonta) or a long branched tube (as in Mytilus). Typically, one end of the tubule opens in the pericardial
cavity via a nephrostome (see earthworm excretion for a general discussion of invertebrate excretion) (after all
the pericardial cavity corresponds to the coelom cavity). The other end of the tubule opens into the mantle
cavity via a nephridiopore. The auricles and glands in the pericardium (pericardial glands) contain a porous
epithelium (composed of podocytes resembling podocytes in the mammalian kidney) and it is thought that
ultrafiltration occurs across these membranes to remove excess water and waste materials.
Nervous System
the nervous system is a paired structure, with pairs of ganglia and nerve cords. The 'brain' consists of a pair of
cerebropleural ganglia (fused cerebral and pleural ganglia) one on each side of the oesophagus, connected
by a dorsal commissure (connecting nerve tract). Two pairs of longitudinal nerve cords extend from these
ganglia. One pair innervates the visceral mass, terminating in visceral ganglia which innervate the posterior
adductor muscle, the siphons (if present) and the internal viscera and gills. The other pair terminate in pedal
ganglia in the foot which innervate the anterior adductor muscle and the foot. The cerebral ganglia directly
innervate the anterior adductor muscle and the palps.
The foot contains one pair of statocysts, which are near to or within the pedal ganglia but innervated by the
cerebral ganglia. These are organs of equilibrium (balance). Eyes may be present. Giant clams have thousands
of simple eyes or ocelli, which are compound eyes in ark shells. Some bivalves have eyes on the head, though
this is not typical in bivalves. The mode of operation of scallop eyes has already been discussed. The
osphradium is a patch of sensory epithelium beneath the posterior adductor muscle which is thought to
monitor the water passing through the mantle cavity.
Reproduction and Development
Most bivalves are dioecious (with separate sexes) but some are hermaphrodites (e.g. some scallops). There is
a pair of gonads, but these are often more-or-less fused into a single mass. In hermaphrodite scallops the
ventral region is the ovary, the dorsal the testis. The gonads open, via simple gonoducts, either into the
nephridium, into which the gametes are shed to exit through the nephridiopores, or else the gonoducts open
directly into the mantle cavity.
In some forms the gender changes during the course of development. In the edible oyster (Ostrea edulis) the
gender periodically switches in a single individual. The gametes are shed into the surrounding water and
fertilisation is generally external, although the female may draw in spermatozoa from the surrounding water, in
the inhalent current, to fertilise the eggs in the suprabranchial cavity (region of the mantle cavity above the gills)
where the eggs may be brooded. The eggs hatch into a trochophore larva which develops into a veliger larva
with two valves. The veliger may feed planktonically or it may be nonfeeding (lechitotrophic, that is deriving
nourishment by digesting yolk reserves) before metamorphosing into a small adult. In most freshwater bivalves
development is direct, e.g. eggs are brooded in marsupial sacs between the gill lamellae in freshwater clams
and hatch into young adult clams.
In some bivalves the veliger is modified to lead a parasitic larval life on fish. One such type of modified veliger is
the glochidium which attaches to the gills, fins or body surface of a host fish via a hook on each valve and an
adhesive thread borne on the rudimentary foot. Some clams release glochidia embedded in wormlike coloured
masses. The host fish eats this mass whilst the glochidia attach to its gills. The fish serves to help disperse the
larva. The host tissues form a cyst around the glochidium and phagocytic cells in the larva's mantle ingest the
host tissues growing around it, deriving nourishment. After 10 to 30 days the larva breaks out of the cyst, sinks
to the bottom and burrows into the sediment to continue its development into adulthood. Such a parasitic life
style requires the production of large numbers of parasites, for most larva will never locate a suitable host.
Some freshwater mussels produce about 3 million glochidia.
Bivalves live up to 150 years (possibly more in some cases) though most live between 2 and 30 years. The
scallop Placopecten requires ten years to reach its maximum diameter of 15 cm. The growth rings on the valves
have already been discussed.
Ecology
Many bivalves are soft-bottom dwellers, burrowing in the sediment. Others live attached to solid surfaces either
via byssal threads (which form the anchor or byssus) or by the cementing of one valve to the substrate. Byssal
threads are very strong protein fibres secreted by the byssus gland with the help of the foot (the thread lies in a
grrove on the pedal foot during secretion and pedal glands contribute to its formation). After being secreted and
anchored, the foot withdrawals, leaving the byssal threads connected to the byssal gland via a strong stem. the
byssal fibres tan and harden in the air. The bivalve can pull on its anchoring byssus with the byssus retractor
muscle.
Some bivalves bore into rock or coral. The giant clam Tridacna gigas (which can reach 1.37 m in diameter)
bores into coral. Blood sinuses in its mantle contain photosynthetic zooxanthellae, which utilise sunlight to
produce food for themselves and the host clam via photosynthesis. The tissues are coloured blue, green, red,
violet or brown by pigments which may act as screening pigments to shield the zooxanthellae from excess
sunlight. Boring bivalves dwell in a permanent burrow, attached to one side via byssal threads or by a sucker on
the foot. Drilling is mechanical, the anterior ends of the valves slowly scraping away the substrate (they may
have serrated edges to assist in this) but can also be chemical in which secretions soften shell or coral to
facilitate mechanical drilling. In soft rocks (shale) drilling can exceed 1 cm depth a month. Shipworms drill into
wood, such as mangrove roots, timber washed out to sea and ship hulls. They may burrow up to 2 m into the
wood and eat the sawdust produced, which is digested with the help of enzymes secreted by symbiotic bacteria.
Some bivalves live as commensals with echinoderms or crustaceans. At least one form is parasitic in sea
cucumber guts.
Above and left: sections through the foot of the bivalve Unio (a
freshwater mussel) showing the glandular epithelium and the
bundles of muscle fibres, some of which criss-cross from one
side of the foot to the other.
freshwater mussel) showing the glandular epithelium and the
bundles of muscle fibres, some of which criss-cross from one
side of the foot to the other.
The scallop eye also contains a lens in front of the reflector, but this lends diffracts light only weakly and is
thought to possibly correct for spherical aberration. Spherical aberration is the tendency for the edges of a lens or
reflective dish to focus light imprecisely, leading to blurring. By directing the light onto the central region of the
reflector, the lens reduced this aberration.
thought to possibly correct for spherical aberration. Spherical aberration is the tendency for the edges of a lens or
reflective dish to focus light imprecisely, leading to blurring. By directing the light onto the central region of the
reflector, the lens reduced this aberration.
The Foot and Visceral Mass
The foot is situated anteriorly and is a bladelike, muscular extension of the body. Many bivalves use their foot in burrowing.
The foot is extended down into the sediment, it then expands at its distal end to anchor the bivalve as the adductor muscles
contract to decrease the thickness of the bivalve by drawing the valves closer together and pedal retractor muscles
shorten the foot, thus pulling the bivalve down into the sediment. Foot extension is accompanied by pedal protractor
muscles and increased blood pressure.
The foot is situated anteriorly and is a bladelike, muscular extension of the body. Many bivalves use their foot in burrowing.
The foot is extended down into the sediment, it then expands at its distal end to anchor the bivalve as the adductor muscles
contract to decrease the thickness of the bivalve by drawing the valves closer together and pedal retractor muscles
shorten the foot, thus pulling the bivalve down into the sediment. Foot extension is accompanied by pedal protractor
muscles and increased blood pressure.
The Shell
Typically the valves are more-or-less oval and convex. There is a dorsal, rounded, protuberance on each valve, called the
umbo, which is the oldest part of the shell. The hinge ligament connects the two valves and this is made of protein and
elastically opens the shell and is opposed by the anterior and posterior adductor muscles. There are teeth or ridges on the
hinge line which fit into sockets on the opposite valve and serve to prevent the valves slipping sideways relative to
one-another.
The shell consists of an outer layer, the periostracum (which also covers the outside of the hinge ligament) and 2-4 inner
calcareous layers of calcium carbonate, in the form of aragonite and sometimes also calcite crystals arranged in prisms or
staggered bricks (the staggering increases the strength of bonding between the crystals, the same principle being used in
brick walls). Calcium carbonate is soluble in acidified water and the periostracum protects the shell from dissolution and also
forms a tight seal when the valves close. Ribbing and corrugation may be present (e.g. in cockles and scallops) and this
increases the mechanical strength of the shell. Shell size may vary from 2 mm to over 1 metre in the giant clam (Tridacna).
The giant clam may weigh over 1100 kg.
A section through the shell reveals various growth lines, some of which are usually visible on the outer surface of the shell.
The more closely-spaced lines are due to etching of the shell by respiratory acids when the shell is closed and so record
daily tides and semilunar rhythms (in which the shell closes when the tide goes out). Annual growth increments also produce
annual rings, generally visible on the outer surface of the shell, allowing the age of the bivalve to be estimated. These may
be caused by winter breaks in the perisotracum, when growth temporarily ceases. Storms may, however, produce additional
lines.
Typically the valves are more-or-less oval and convex. There is a dorsal, rounded, protuberance on each valve, called the
umbo, which is the oldest part of the shell. The hinge ligament connects the two valves and this is made of protein and
elastically opens the shell and is opposed by the anterior and posterior adductor muscles. There are teeth or ridges on the
hinge line which fit into sockets on the opposite valve and serve to prevent the valves slipping sideways relative to
one-another.
The shell consists of an outer layer, the periostracum (which also covers the outside of the hinge ligament) and 2-4 inner
calcareous layers of calcium carbonate, in the form of aragonite and sometimes also calcite crystals arranged in prisms or
staggered bricks (the staggering increases the strength of bonding between the crystals, the same principle being used in
brick walls). Calcium carbonate is soluble in acidified water and the periostracum protects the shell from dissolution and also
forms a tight seal when the valves close. Ribbing and corrugation may be present (e.g. in cockles and scallops) and this
increases the mechanical strength of the shell. Shell size may vary from 2 mm to over 1 metre in the giant clam (Tridacna).
The giant clam may weigh over 1100 kg.
A section through the shell reveals various growth lines, some of which are usually visible on the outer surface of the shell.
The more closely-spaced lines are due to etching of the shell by respiratory acids when the shell is closed and so record
daily tides and semilunar rhythms (in which the shell closes when the tide goes out). Annual growth increments also produce
annual rings, generally visible on the outer surface of the shell, allowing the age of the bivalve to be estimated. These may
be caused by winter breaks in the perisotracum, when growth temporarily ceases. Storms may, however, produce additional
lines.
The Mantle
A sheet of tissue, extending from the main body, covers the inner surface of each valve and constitute the mantle. In
between these sheets and the main body (visceral mass) lies the mantle cavity containing the gill(s). The mantle, which is
covered by the epithelium of the body wall, contains muscle fibres. A semicircular arc of mantle muscle fibres attach the
mantle lobe to the shell, which leaves a visible scar on many bivalve valves, called the pallial line. The mantle is responsible
for secreting the shell and also has a sensory function (e.g. bearing eyes, guard tentacles and sensory tentacles in scallops).
Particles ('seeds') which get trapped between the mantle and shell become coated in secreted shell material (whether this is
a deliberate response to irritation or just a consequence of the mantle secreting new shell materials I am not sure). This
forms a pearl. If the particle gets rolled about then the pearl becomes spheroidal. Otherwise the pearl may fuse to the shell.
All bivalves produce pearls in this way, but only some coat it in shiny or nacreous material (nacre) and these are of
commercial value. Pearls vary from whitish to pink in colour. Pearl oysters (Pinctada spp.) have 'seeds' deliberately impanted
in them and these are farmed after 3 years of growth.
A sheet of tissue, extending from the main body, covers the inner surface of each valve and constitute the mantle. In
between these sheets and the main body (visceral mass) lies the mantle cavity containing the gill(s). The mantle, which is
covered by the epithelium of the body wall, contains muscle fibres. A semicircular arc of mantle muscle fibres attach the
mantle lobe to the shell, which leaves a visible scar on many bivalve valves, called the pallial line. The mantle is responsible
for secreting the shell and also has a sensory function (e.g. bearing eyes, guard tentacles and sensory tentacles in scallops).
Particles ('seeds') which get trapped between the mantle and shell become coated in secreted shell material (whether this is
a deliberate response to irritation or just a consequence of the mantle secreting new shell materials I am not sure). This
forms a pearl. If the particle gets rolled about then the pearl becomes spheroidal. Otherwise the pearl may fuse to the shell.
All bivalves produce pearls in this way, but only some coat it in shiny or nacreous material (nacre) and these are of
commercial value. Pearls vary from whitish to pink in colour. Pearl oysters (Pinctada spp.) have 'seeds' deliberately impanted
in them and these are farmed after 3 years of growth.
Respiration and Feeding
Some bivalves (of a type called protobranchs) feed by extending the palpal tentacles into the sediment. Beating cilia on
the tentacles drive water currents towards the pair of palps which lies at the base of each tentacle. Visibly there is little
more to the head in bivalves. The mouth sits between the bases of the palps. These currents drive particles of sediment to
the palps where more cilia drive them over the surface of the palps where they cross a series of ridges. Lighter food
particles are carried across from the crest of one ridge to another and in through the mouth, and these particles consist
mostly of organic matter. Heavier particles, such as sand particles, fall between the ridges where cilia in these troughs drive
the particles off the tips of the palps to be rejected.
Cilia on the gills also drive water currents over the gills, where the oxygen can be absorbed and carbon dioxide removed.
The respiring and feeding bivalve sits (perhaps in a burrow) with its valves agape. Water enters through the posterior
ventral shell gape to be carried up through the gills and finally to exit through the posterior dorsal shell gape. Generally, a
bivalve will sit in its burrow head down (foot down) and water will thus be drawn from the surface through and out of the
posterior end. The gills consist of a series of filaments which may be modified and grouped into W-shaped tubes with the
ascending and descending limbs aligned to form sheets or lamellae (an aligned column of Ws forming four such sheets).
Most bivalves have this type of gill and are called lamellibranchs. Protobranches have a more ancestral type of gill, called
a protobranch gill, in which the gills consist of a series of V-shaped filaments. The lamellibranch gill is an adaptation for
filter-feeding, whereas protobranchs are generally deposit feeders, as desrcibed above. The arrangement of the three
principle types of lamellibranch gill and their role in filter-feeding will now be excplained.
In bivalves such as Pecten (a scallop) and Mytilis (mussel) these filaments are more-or-less separate, but adjacent filaments
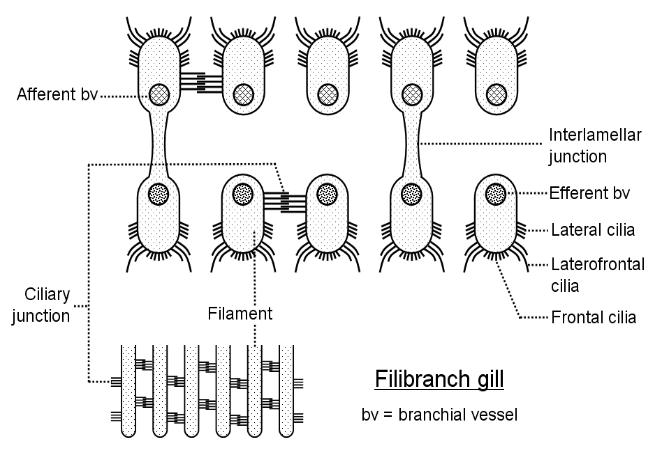
may be joined by interlocking cilia (ciliary junction) which maintains the correct spacing between the filaments.
Interlamellar junctions which are struts of connective tissue join neighbouring limbs within each W. This type of gill is the
filobranch gill (lamellibranch gill type 1).
Some bivalves (of a type called protobranchs) feed by extending the palpal tentacles into the sediment. Beating cilia on
the tentacles drive water currents towards the pair of palps which lies at the base of each tentacle. Visibly there is little
more to the head in bivalves. The mouth sits between the bases of the palps. These currents drive particles of sediment to
the palps where more cilia drive them over the surface of the palps where they cross a series of ridges. Lighter food
particles are carried across from the crest of one ridge to another and in through the mouth, and these particles consist
mostly of organic matter. Heavier particles, such as sand particles, fall between the ridges where cilia in these troughs drive
the particles off the tips of the palps to be rejected.
Cilia on the gills also drive water currents over the gills, where the oxygen can be absorbed and carbon dioxide removed.
The respiring and feeding bivalve sits (perhaps in a burrow) with its valves agape. Water enters through the posterior
ventral shell gape to be carried up through the gills and finally to exit through the posterior dorsal shell gape. Generally, a
bivalve will sit in its burrow head down (foot down) and water will thus be drawn from the surface through and out of the
posterior end. The gills consist of a series of filaments which may be modified and grouped into W-shaped tubes with the
ascending and descending limbs aligned to form sheets or lamellae (an aligned column of Ws forming four such sheets).
Most bivalves have this type of gill and are called lamellibranchs. Protobranches have a more ancestral type of gill, called
a protobranch gill, in which the gills consist of a series of V-shaped filaments. The lamellibranch gill is an adaptation for
filter-feeding, whereas protobranchs are generally deposit feeders, as desrcibed above. The arrangement of the three
principle types of lamellibranch gill and their role in filter-feeding will now be excplained.
In bivalves such as Pecten (a scallop) and Mytilis (mussel) these filaments are more-or-less separate, but adjacent filaments
may be joined by interlocking cilia (ciliary junction) which maintains the correct spacing between the filaments.
Interlamellar junctions which are struts of connective tissue join neighbouring limbs within each W. This type of gill is the
filobranch gill (lamellibranch gill type 1).
Above: a pseudolamellibranch gill, as occurs in oysters and pen shells (lamellibranch gill type 2). The arrangement of
each filament into a W creates what is effectively a pair of gills or demibranchs ('half-gills'): an inner V-shaped demibranch
near to the visceral mass and an outer v-shaped demibranch attached to the mantle. In this type of gill, the junctions
between adjacent filaments are not made from interlocking cilia, but rather consist of actual bridges of connective tissue.
These are the interfilamentous junctions or interfilamentous connectives. This arranges the gill into properly connected
lamellae.
each filament into a W creates what is effectively a pair of gills or demibranchs ('half-gills'): an inner V-shaped demibranch
near to the visceral mass and an outer v-shaped demibranch attached to the mantle. In this type of gill, the junctions
between adjacent filaments are not made from interlocking cilia, but rather consist of actual bridges of connective tissue.
These are the interfilamentous junctions or interfilamentous connectives. This arranges the gill into properly connected
lamellae.
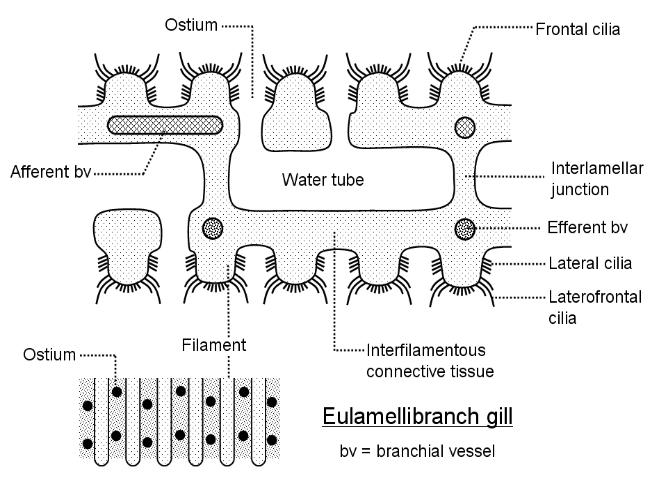
The third major type of lamellibranch gill is the eulamellibranch gill, in which the interfilamentous junctions become so
extensive that the filaments form four true lamellae or sheets with the adjacent filaments reduced to ciliated ridges with
sheets of connective tissue between them, perforated by pores or ostia. The spaces between the lamellae then become
water tubes. Remember that lamellibranchs generally use their gills in filter-feeding as well as for respiration.
In all three cases, rows of lateral cilia along the adjacent edges of the filaments generate the main water currents, drawing
water in between the filaments (or through the ostia in eulamellibranch gills). Water then flows in the spaces between the
lamellae, along the axis of the gill and out at the end of the gill. This current is for gas exchange.
In many bivalves, however, the gills have become modified for use in filter-feeding as well as for respiration. Packing in
more filaments and lengthening them (and packaging them into W-shapes) is part of the adaptation for this. Modified cilia,
called laterofrontal cilia, form stiff bundles called cirri (singular cirrus) which form a mesh to strain out particles from the
water passing between each pair of adjacent filaments. Frontal cilia then convey these particles to the vertices or corners
of each W-shaped filament where they enter food grooves which transport the food particles along the gill, from filament to
filament, towards the palps and hence to the mouth. A W has five such channels (one at each of three angles and one on
each of the two V-shaped points or apices) however, in many forms some of these channels do not function as food
grooves and in some cases only the two apical channels are used. this complex arrangement of gill parts is illustrated below.
extensive that the filaments form four true lamellae or sheets with the adjacent filaments reduced to ciliated ridges with
sheets of connective tissue between them, perforated by pores or ostia. The spaces between the lamellae then become
water tubes. Remember that lamellibranchs generally use their gills in filter-feeding as well as for respiration.
In all three cases, rows of lateral cilia along the adjacent edges of the filaments generate the main water currents, drawing
water in between the filaments (or through the ostia in eulamellibranch gills). Water then flows in the spaces between the
lamellae, along the axis of the gill and out at the end of the gill. This current is for gas exchange.
In many bivalves, however, the gills have become modified for use in filter-feeding as well as for respiration. Packing in
more filaments and lengthening them (and packaging them into W-shapes) is part of the adaptation for this. Modified cilia,
called laterofrontal cilia, form stiff bundles called cirri (singular cirrus) which form a mesh to strain out particles from the
water passing between each pair of adjacent filaments. Frontal cilia then convey these particles to the vertices or corners
of each W-shaped filament where they enter food grooves which transport the food particles along the gill, from filament to
filament, towards the palps and hence to the mouth. A W has five such channels (one at each of three angles and one on
each of the two V-shaped points or apices) however, in many forms some of these channels do not function as food
grooves and in some cases only the two apical channels are used. this complex arrangement of gill parts is illustrated below.
Taking a horizontal section through two adjacent lamellae, as
indicated on the left, and viewing the gill filaments from their cut
ends we see something like that illustrated in the two diagrams
below, for a filobranch gill with ciliary junctions and for a
eulamellibranch gill.
indicated on the left, and viewing the gill filaments from their cut
ends we see something like that illustrated in the two diagrams
below, for a filobranch gill with ciliary junctions and for a
eulamellibranch gill.
Above: a section through a filobranch gill. Note the ciliary junctions (interfilamentous junctions) and the
connective tissue junctions between each pair of lamellae within the same filament. comparing this to the
3D model shown above should help you visualise the arrangement. The inset (bottom left) shows one
lamella in surface view.
connective tissue junctions between each pair of lamellae within the same filament. comparing this to the
3D model shown above should help you visualise the arrangement. The inset (bottom left) shows one
lamella in surface view.
Above: a section through a eulamellibranch gill. This time, extensive sheets of connective tissue,
perforated by ostia, connect adjacent filaments (interfilamentous connective tissue) such that each
lamella when seen in surface view (inset, bottom left) appears like a solid sheet with ridges and pores.
perforated by ostia, connect adjacent filaments (interfilamentous connective tissue) such that each
lamella when seen in surface view (inset, bottom left) appears like a solid sheet with ridges and pores.
The visceral mass is the main body of the animal, containing the gut and vascular systems.
Article last updated: 27/12/2014