| The Icefish |



Above: the icefish Chaenocephalus aceratus. This fish is about 50 cm long. Note the relatively large head and
small tail. This fish is an ambush predator, resting on the bottom by propping itself up on its strong pelvic fins
(the two fins coming from the chest underneath).
Life in extremes!
The icefishes (chaenicthyids) are a bizarre group of fish that live in the Antarctic Ocean. They are
extremophiles (organisms that thrive in extreme conditions) adapted to cold environments. Specifically they
are cryophiles ('cold-liking' organisms) that live in Antarctic waters at a temperature of about minus 1.7C
(-1.9C to + 3C). Although some fish are endothermic (warm-blooded, meaning they generate their own body
heat) most are exothermic or poikilothermic ('cold-blooded') obtaining almost all of their heat from the outside
environment. Living in water that conducts heat well, this means that their body temperature is the same as
the surrounding water. In other words ice fish have body temperatures just a fraction of a degree above minus
2 degrees C. If warmed to +6C then they die of heat-sickness! Even a refrigerator at +4C would be
uncomfortably hot for an ice fish!
In addition to the cold, Antarctic waters are dark and deep. During the long Antarctic winter there is very little
light and in the summer sea ice still screens out much sunlight. The Antarctic continental shelf is also deeper
than for any other continent, so organisms living on the seabed are at considerable depth. For these reasons,
Antarctic fish share some of the adaptations of the strange deep-ocean fish, though to a lesser degree.
Like many Antarctic fish, icefishes may produce biological antifreeze - glycoproteins in body fluids that
surround ice crystals and prevent them from growing. Sea water at -2C is supercooled - it does not freeze
easily because it is salty, however, when an animal that is supercooled touches ice, sudden and catastrophic
freezing may occur. Fish that swim near the surface may bump into ice and have particularly effective
antifreeze. Fish may also swallow ice as they drink sea water and this ice must be prevented from freezing the
fish! Chaenocephaluis aceratus generally stays near the sea bed, however, ice sometimes penetrates down
and this animal does have some antifreeze glycoproteins in its fluids, though not as much as some more
active surface swimmers.
Colourless blood!
There are some 16 species of icefish which share their environment with other fish and other organisms -
despite the extreme cold the antarctic Ocean is a very rich ecosystem. However, the ice fish are peculiar in
lacking red blood cells (RBCs or erythrocytes) and the red oxygen-carrying pigment haemoglobin! Most, like
Chaenocephalus aceratus, have no haemoglobin at all! This is surprising since haemoglobin carries 90% of
the oxygen in the bloodstream of a typical vertebrate, the remaining 10% is oxygen dissolve din physical
solution in the water of the blood. It is very hard indeed to imagine how a vertebrate can survive without
haemoglobin. The blood of icefish is colourless and slightly cloudy or turbid (white). (Actually a few
'rudimentary' erythrocytes do occur in th blood of some icefishes, but these carry no haemoglobin at all). They
are hard to detect, since their bodies are translucent (rather like ice!) save for a few vertical lines of dark
spots. This camouflages them and makes it easier for them to ambush prey and to hide from predators.
What we shall endeavour to explain in this article, is how these bizarre creatures survive without haemoglobin.
How do they get enough oxygen to their tissues and active muscles?
The discussion in this article is quite technical and a useful preliminary (for those not familiar) would be read
the article on respiration and mitochondria first and also the article on diffusion.
Why no haemoglobin?
The problem with low temperatures is that fluids become more viscous 9sticky or treacle-like) when they are
cold. The viscosity of water doubles when the temperature is lowered from 30C to 0C. The viscosity of blood
more than doubles, but blood is already three times more viscous than water, so blood at 0C is 4 times more
viscous than water at 0C and 8 times more viscous than water at 30C. This highly viscous blood creates
problems for the heart which has to squeeze this thick liquid through tiny blood capillaries (capillaries are the
smallest blood vessels that form fine branching networks to supply the tissues). The viscosity of blood is due
mostly to proteins in the plasma (the plasma is the fluid component of blood) and blood cells. In all other
vertebrates, including humans, red blood cells account for the vast majority of blood cells (white blood cells
are far fewer in number). Haematocrit is the fraction of blood volume taken-up by red blood cells and is about
40% in humans. A similar thing happens to athletes who extract some of their blood, remove the red blood
cells and then re-inject these cells into their veins prior to a race, in order to increase the amount of oxygen
their blood can carry: this greatly increases their blood viscosity and places strain on the heart which may lead
to heart failure. This also dangerously increases blood pressure as the heart has to exert more pressure to
keep the blood circulating. Antarctic fish have countered this problem by reducing their haematocrit, and the
icefish have gone to the extreme by reducing their haematocrit to essentially zero.
Icefish have no oxygen carrying pigment in their blood at all. (Even haemoglobin free in the plasma would give
the blood high viscosity). They rely on the 10% of the oxygen that normally dissolves into the water of the
blood (oxygen is moderately soluble in water) in a physical solution (that is the oxygen does not react
chemically with the water, as does say salt when salt dissolves, but some oxygen molecules simply get caught
between the water molecules, a mechanism that does not dissolve great quantities). Thus, they simply carry
only 10% of the oxygen in their blood that they could do if they had haemoglobin.
How do icefish survive with so little oxygen in their colourless blood?
Effect of cold on oxygen transport
The cold temperatures compensate to a small degree, since oxygen is more soluble in cold water than it is in
warm water (solids like salt and sugar dissolve better in warm water, but gases are different). However, this is
not enough, especially as oxygen must enter and move around inside a muscle cell by diffusion, and diffusion
is slower in the cold. In fact what really matters is the permeability of tissues to oxygen and permeability = rate
of diffusion x solubility, and the increased solubility of oxygen in cold water actually compensates for the slower
rate of diffusion of oxygen in cold water.
A lazy lifestyle?
Part of the solution lies in lifestyle. Icefish are generally not very active. For example, Chaenocephalus
aceratus sits on the seabed, waiting in ambush for its prey, propped up on its two pelvic fins like a bipod.
When it swims it uses the pectoral fins (the pair of broad fins on the sides of its chest) which trace a
figure-of-eight through the water. During the power stroke the pectoral fins are vertical so as to drag water
backwards and downwards to create thrust and then during the recovery stroke the fins move forwards narrow
edge first, slicing through the water with minimal effort. (This form of swimming is called drag-based labriform
swimming). This graceful swimming is quite slow and relatively effortless. The tail simply steers the fish like a
rudder and only forcefully propels the fish for brief dashes, as when pouncing on prey or escaping from a
predator. Thus, the tail is relatively small. In more active fish the large tail is the main motor. However, this
mode of life only modestly reduces the oxygen needs of icefish and some icefish are more active pelagic fish,
staying afloat by constant swimming in the surface waters. Thus, this is still not the whole answer. Icefish
muscle is also quite watery, having a reduced protein content, making it lighter and more buoyant, which
reduces the amount of work needed to swim. Antarctic fish generally have large lipid sacs in the body (outside
the muscles) which also increase buoyancy.
Fish have two main types of muscle cell: red aerobic muscle that can exercise slowly for prolonged periods
and anaerobic white muscle which is stronger and faster but fatigues quickly and is used for sprinting. The
pectoral fins of icefish are controlled by 6 pairs of muscles which are aerobic muscles. The tail is mostly
anaerobic white muscle, for quick dashes, though a small amount of red muscle in the tail steers the tail as a
rudder. When dashing to catch prey, the white muscles in the tail, especially, will build-up lactic acid by
anaerobic respiration. After catching prey, the large mouth allows easy ingestion and then the icefish can rest
and digest its food while it slowly restores its oxygen levels and uses oxygen to metabolism the lactic acid.
Carrying more oxygen without haemoglobin
If icefish blood can only carry 10% of the oxygen per ml of blood that a red blooded fish can carry, then one
solution is to have more blood! The blood volume of the icefish is large at about 2-4 times larger than for
a red-blooded fish of the same size. This alone means that 10% becomes 20-40%. Additionally icefish have
more blood capillaries in their skin and fins which can absorb oxygen direct from the sea water. About
30-40% of the oxygen is absorbed across the skin, which is totally devoid of scales, making it thin to allow
oxygen to diffuse across it more easily. The gills absorb the rest and are of a normal surface area. However,
these mechanisms are still not the whole answer. Most of this extra blood must be stored in the capillaries,
since this is where most of the blood volume resides in vertebrates. Delivering this blood requires a larger
pump and the heart has a very large stroke volume (the volume of blood pumped in a single heartbeat)
6-15 times higher than for a red-blooded fish of similar size. In fact the icefish heart has a capacity similar to
that of a vertebrate heart (getting about on dry land can be hard work). The heart only beats 15 times a
minute (compare 60-80 bpm for a typical human) and does not have to beat very hard as the blood
pressure is low - the low viscosity blood slips easily through the unusually large capillaries which are
actually more than double the normal diameter (with a radius of 4.5 micrometres, compared to ). Thus a large
volume of blood flows quickly through the tissues. The blood vessels (lacunae) in the gills are also
large, so blood flows rapidly through the gills, so that more oxygen is picked up. In animals with red blood cells,
the opposite is generally true - capillaries are tiny, forcing red blood cells to squeeze slowly through them,
allowing enough time for haemoglobin to absorb oxygen in the gills/lungs and to deposit it in tissues/exercising
muscles.
Muscle structure
To get more answers, scientists have studied the structure of icefish muscles under the light and electron
microscopes,a nd also the nature of the blood vessels supplying them. One key parameter is the blood
capillary to muscle fibre ratio (C:F). Consider a cross-section through a small piece of muscle as shown
diagrammatically below:
small tail. This fish is an ambush predator, resting on the bottom by propping itself up on its strong pelvic fins
(the two fins coming from the chest underneath).
Life in extremes!
The icefishes (chaenicthyids) are a bizarre group of fish that live in the Antarctic Ocean. They are
extremophiles (organisms that thrive in extreme conditions) adapted to cold environments. Specifically they
are cryophiles ('cold-liking' organisms) that live in Antarctic waters at a temperature of about minus 1.7C
(-1.9C to + 3C). Although some fish are endothermic (warm-blooded, meaning they generate their own body
heat) most are exothermic or poikilothermic ('cold-blooded') obtaining almost all of their heat from the outside
environment. Living in water that conducts heat well, this means that their body temperature is the same as
the surrounding water. In other words ice fish have body temperatures just a fraction of a degree above minus
2 degrees C. If warmed to +6C then they die of heat-sickness! Even a refrigerator at +4C would be
uncomfortably hot for an ice fish!
In addition to the cold, Antarctic waters are dark and deep. During the long Antarctic winter there is very little
light and in the summer sea ice still screens out much sunlight. The Antarctic continental shelf is also deeper
than for any other continent, so organisms living on the seabed are at considerable depth. For these reasons,
Antarctic fish share some of the adaptations of the strange deep-ocean fish, though to a lesser degree.
Like many Antarctic fish, icefishes may produce biological antifreeze - glycoproteins in body fluids that
surround ice crystals and prevent them from growing. Sea water at -2C is supercooled - it does not freeze
easily because it is salty, however, when an animal that is supercooled touches ice, sudden and catastrophic
freezing may occur. Fish that swim near the surface may bump into ice and have particularly effective
antifreeze. Fish may also swallow ice as they drink sea water and this ice must be prevented from freezing the
fish! Chaenocephaluis aceratus generally stays near the sea bed, however, ice sometimes penetrates down
and this animal does have some antifreeze glycoproteins in its fluids, though not as much as some more
active surface swimmers.
Colourless blood!
There are some 16 species of icefish which share their environment with other fish and other organisms -
despite the extreme cold the antarctic Ocean is a very rich ecosystem. However, the ice fish are peculiar in
lacking red blood cells (RBCs or erythrocytes) and the red oxygen-carrying pigment haemoglobin! Most, like
Chaenocephalus aceratus, have no haemoglobin at all! This is surprising since haemoglobin carries 90% of
the oxygen in the bloodstream of a typical vertebrate, the remaining 10% is oxygen dissolve din physical
solution in the water of the blood. It is very hard indeed to imagine how a vertebrate can survive without
haemoglobin. The blood of icefish is colourless and slightly cloudy or turbid (white). (Actually a few
'rudimentary' erythrocytes do occur in th blood of some icefishes, but these carry no haemoglobin at all). They
are hard to detect, since their bodies are translucent (rather like ice!) save for a few vertical lines of dark
spots. This camouflages them and makes it easier for them to ambush prey and to hide from predators.
What we shall endeavour to explain in this article, is how these bizarre creatures survive without haemoglobin.
How do they get enough oxygen to their tissues and active muscles?
The discussion in this article is quite technical and a useful preliminary (for those not familiar) would be read
the article on respiration and mitochondria first and also the article on diffusion.
Why no haemoglobin?
The problem with low temperatures is that fluids become more viscous 9sticky or treacle-like) when they are
cold. The viscosity of water doubles when the temperature is lowered from 30C to 0C. The viscosity of blood
more than doubles, but blood is already three times more viscous than water, so blood at 0C is 4 times more
viscous than water at 0C and 8 times more viscous than water at 30C. This highly viscous blood creates
problems for the heart which has to squeeze this thick liquid through tiny blood capillaries (capillaries are the
smallest blood vessels that form fine branching networks to supply the tissues). The viscosity of blood is due
mostly to proteins in the plasma (the plasma is the fluid component of blood) and blood cells. In all other
vertebrates, including humans, red blood cells account for the vast majority of blood cells (white blood cells
are far fewer in number). Haematocrit is the fraction of blood volume taken-up by red blood cells and is about
40% in humans. A similar thing happens to athletes who extract some of their blood, remove the red blood
cells and then re-inject these cells into their veins prior to a race, in order to increase the amount of oxygen
their blood can carry: this greatly increases their blood viscosity and places strain on the heart which may lead
to heart failure. This also dangerously increases blood pressure as the heart has to exert more pressure to
keep the blood circulating. Antarctic fish have countered this problem by reducing their haematocrit, and the
icefish have gone to the extreme by reducing their haematocrit to essentially zero.
Icefish have no oxygen carrying pigment in their blood at all. (Even haemoglobin free in the plasma would give
the blood high viscosity). They rely on the 10% of the oxygen that normally dissolves into the water of the
blood (oxygen is moderately soluble in water) in a physical solution (that is the oxygen does not react
chemically with the water, as does say salt when salt dissolves, but some oxygen molecules simply get caught
between the water molecules, a mechanism that does not dissolve great quantities). Thus, they simply carry
only 10% of the oxygen in their blood that they could do if they had haemoglobin.
How do icefish survive with so little oxygen in their colourless blood?
Effect of cold on oxygen transport
The cold temperatures compensate to a small degree, since oxygen is more soluble in cold water than it is in
warm water (solids like salt and sugar dissolve better in warm water, but gases are different). However, this is
not enough, especially as oxygen must enter and move around inside a muscle cell by diffusion, and diffusion
is slower in the cold. In fact what really matters is the permeability of tissues to oxygen and permeability = rate
of diffusion x solubility, and the increased solubility of oxygen in cold water actually compensates for the slower
rate of diffusion of oxygen in cold water.
A lazy lifestyle?
Part of the solution lies in lifestyle. Icefish are generally not very active. For example, Chaenocephalus
aceratus sits on the seabed, waiting in ambush for its prey, propped up on its two pelvic fins like a bipod.
When it swims it uses the pectoral fins (the pair of broad fins on the sides of its chest) which trace a
figure-of-eight through the water. During the power stroke the pectoral fins are vertical so as to drag water
backwards and downwards to create thrust and then during the recovery stroke the fins move forwards narrow
edge first, slicing through the water with minimal effort. (This form of swimming is called drag-based labriform
swimming). This graceful swimming is quite slow and relatively effortless. The tail simply steers the fish like a
rudder and only forcefully propels the fish for brief dashes, as when pouncing on prey or escaping from a
predator. Thus, the tail is relatively small. In more active fish the large tail is the main motor. However, this
mode of life only modestly reduces the oxygen needs of icefish and some icefish are more active pelagic fish,
staying afloat by constant swimming in the surface waters. Thus, this is still not the whole answer. Icefish
muscle is also quite watery, having a reduced protein content, making it lighter and more buoyant, which
reduces the amount of work needed to swim. Antarctic fish generally have large lipid sacs in the body (outside
the muscles) which also increase buoyancy.
Fish have two main types of muscle cell: red aerobic muscle that can exercise slowly for prolonged periods
and anaerobic white muscle which is stronger and faster but fatigues quickly and is used for sprinting. The
pectoral fins of icefish are controlled by 6 pairs of muscles which are aerobic muscles. The tail is mostly
anaerobic white muscle, for quick dashes, though a small amount of red muscle in the tail steers the tail as a
rudder. When dashing to catch prey, the white muscles in the tail, especially, will build-up lactic acid by
anaerobic respiration. After catching prey, the large mouth allows easy ingestion and then the icefish can rest
and digest its food while it slowly restores its oxygen levels and uses oxygen to metabolism the lactic acid.
Carrying more oxygen without haemoglobin
If icefish blood can only carry 10% of the oxygen per ml of blood that a red blooded fish can carry, then one
solution is to have more blood! The blood volume of the icefish is large at about 2-4 times larger than for
a red-blooded fish of the same size. This alone means that 10% becomes 20-40%. Additionally icefish have
more blood capillaries in their skin and fins which can absorb oxygen direct from the sea water. About
30-40% of the oxygen is absorbed across the skin, which is totally devoid of scales, making it thin to allow
oxygen to diffuse across it more easily. The gills absorb the rest and are of a normal surface area. However,
these mechanisms are still not the whole answer. Most of this extra blood must be stored in the capillaries,
since this is where most of the blood volume resides in vertebrates. Delivering this blood requires a larger
pump and the heart has a very large stroke volume (the volume of blood pumped in a single heartbeat)
6-15 times higher than for a red-blooded fish of similar size. In fact the icefish heart has a capacity similar to
that of a vertebrate heart (getting about on dry land can be hard work). The heart only beats 15 times a
minute (compare 60-80 bpm for a typical human) and does not have to beat very hard as the blood
pressure is low - the low viscosity blood slips easily through the unusually large capillaries which are
actually more than double the normal diameter (with a radius of 4.5 micrometres, compared to ). Thus a large
volume of blood flows quickly through the tissues. The blood vessels (lacunae) in the gills are also
large, so blood flows rapidly through the gills, so that more oxygen is picked up. In animals with red blood cells,
the opposite is generally true - capillaries are tiny, forcing red blood cells to squeeze slowly through them,
allowing enough time for haemoglobin to absorb oxygen in the gills/lungs and to deposit it in tissues/exercising
muscles.
Muscle structure
To get more answers, scientists have studied the structure of icefish muscles under the light and electron
microscopes,a nd also the nature of the blood vessels supplying them. One key parameter is the blood
capillary to muscle fibre ratio (C:F). Consider a cross-section through a small piece of muscle as shown
diagrammatically below:
The higher the C:F the more capillaries there are to each muscle fibre (a muscle fibre = muscle cell) and the
higher the amount of oxygen delivered to the muscle cell. In the diagram above there are 12 capillaries and 7
muscle fibres, giving a C:F of 1.71. In Chaenocephalus aceratus the (6 pairs of) muscles controlling the
swimming pectoral muscles are aerobic and so require a constant supply of oxygen that is sufficient to meet
demands. However, the C:F of these muscles is only about 1.5 on average (meaning that each muscle fibre is
surrounded by 3 capillaries, since each capillary shares itself between two adjacent muscle fibres on average)
and so is not especially high (though is higher than the C:F of 0.5 to 1 typical of many fish). However, these
capillaries are large and highly sinuous or tortuous, meaning that they undulate across the surface of the
muscle fibre, such that the total length of capillary supplying the fibre is greatly increased.
The diagram below shows three models of a muscle fibre (C:F = 1) which we can model mathematically.
Consider the simplest model first, model A, which assumes that the muscle fibre and capillaries are circular
(and given diameters which are average for the tissue) and the interior of the muscle fibre is assumed uniform.
higher the amount of oxygen delivered to the muscle cell. In the diagram above there are 12 capillaries and 7
muscle fibres, giving a C:F of 1.71. In Chaenocephalus aceratus the (6 pairs of) muscles controlling the
swimming pectoral muscles are aerobic and so require a constant supply of oxygen that is sufficient to meet
demands. However, the C:F of these muscles is only about 1.5 on average (meaning that each muscle fibre is
surrounded by 3 capillaries, since each capillary shares itself between two adjacent muscle fibres on average)
and so is not especially high (though is higher than the C:F of 0.5 to 1 typical of many fish). However, these
capillaries are large and highly sinuous or tortuous, meaning that they undulate across the surface of the
muscle fibre, such that the total length of capillary supplying the fibre is greatly increased.
The diagram below shows three models of a muscle fibre (C:F = 1) which we can model mathematically.
Consider the simplest model first, model A, which assumes that the muscle fibre and capillaries are circular
(and given diameters which are average for the tissue) and the interior of the muscle fibre is assumed uniform.
Let's begin with a crude model in which oxygen is moving out from the capillaries, by diffusion, into and
across the muscle fibre. Ignoring changes over time, we can plot the exponential decrease of oxygen levels
with distance from the capillaries, which is typical of diffusion (see a mathematical example of diffusion). The
3D wire-plot below (left) shows the concentration of oxygen and illustrates its decrease with distance from the
two capillaries as we move into the muscle fibre. On the right is a colour-coded plot (plan view) where the
highest oxygen levels are shown in red (the two capillaries) and the lowest in blue (in the centre of the
muscle fibre). This muscle fibre is not doing too well, the mitochondria in the centre of the muscle have
barely enough oxygen to function!
across the muscle fibre. Ignoring changes over time, we can plot the exponential decrease of oxygen levels
with distance from the capillaries, which is typical of diffusion (see a mathematical example of diffusion). The
3D wire-plot below (left) shows the concentration of oxygen and illustrates its decrease with distance from the
two capillaries as we move into the muscle fibre. On the right is a colour-coded plot (plan view) where the
highest oxygen levels are shown in red (the two capillaries) and the lowest in blue (in the centre of the
muscle fibre). This muscle fibre is not doing too well, the mitochondria in the centre of the muscle have
barely enough oxygen to function!
Note: In this crude model, we have assumed that the oxygen concentration decreases exponentially with
distance from the capillaries. The capillaries are modeled as extended sources (rather than as single points)
however. It incorporates a measure of the rate of diffusion (diffusion coefficient) and increasing the rate of
diffusion of oxygen in the model means that more oxygen reaches the middle of the muscle fibre:
distance from the capillaries. The capillaries are modeled as extended sources (rather than as single points)
however. It incorporates a measure of the rate of diffusion (diffusion coefficient) and increasing the rate of
diffusion of oxygen in the model means that more oxygen reaches the middle of the muscle fibre:
Increasing diffusion still further:
Permeability to Oxygen
This crude model illustrates some key basics. At higher temperatures, for example, in which oxygen diffuses
faster, we might expect more oxygen to reach the middle of the muscle fibre. However, in reality this is slightly
more than compensated for by the increased solubility of oxygen in cold water - the permeability of oxygen
(diffusion coefficient x solubility) actually increases slightly in the cold. Permeability replaces the diffusion
coefficient in our model. Most vertebrate muscle also contains myoglobin, a pink oxygen-binding pigment
which acts as a store of oxygen and also increases transport of oxygen across the muscle fibre. However,
perhaps paradoxically icefish muscles usually have no myoglobin. Chaenocephalus aceratus has traces of
myoglobin in its heart muscle, but none at all in its swimming muscles! Oxygen is more soluble in lipid
(oil) than in water. Muscle fibres are sometimes rich in lipid droplets, which as well as being a store of fuel
may also serve to increase permeability to oxygen, allowing more oxygen to reach the muscle fibre centre.
However, icefish muscle fibres have low lipid content! However, they also have a reduced protein content.
protein significantly impedes oxygen transport across muscle fibres, and the more watery nature of
icefish muscles is expected to significantly increase permeability to oxygen.
Mitochondria and muscle fibre diameter
Distance between the capillaries, or in other words muscle fibre diameter, is also important. In larger fibres
the oxygen has further to travel and this lowers the oxygen concentration in the middle of the fibre (all other
things being equal). Fish grow by increasing muscle fibre diameter, rather than by adding new fibres, so
older and bigger fish have larger muscle fibres and bigger fish tend to rely increasingly less on aerobic
respiration, becoming more anaerobic. Aerobic red muscle fibres are also much smaller than anaerobic white
fibres, since red fibres need faster oxygen delivery. Paradoxically, the red fibres of icefish are unusually
large, larger than is typical for a red-blooded fish of the same size! However, this increase is not chiefly due
to an increase in contractile muscle proteins, nut in mitochondria. Icefish muscles are packed with
mitochondria which account for about 50% of the muscle fibre volume in aerobic swimming muscles.
This proliferation of mitochondria accounts for the large size of the muscle fibres.
This is naively intuitive - it seems as if icefish muscles have more mitochondria to use more oxygen and
make more ATP. However, it is not so simple! For a start, where does the oxygen that the mitochondria need
come from? Detailed analysis has also shown that the mitochondria of icefish have an usually low cristae
density, reduced by 30% (so total cristae surface area is only slightly higher than usual) and enzyme studies
also show that aerobic respiration enzyme activity per gram of mitochondria is about 50% of normal, so the
maximum possible oxygen consumption by the muscle seems normal and is not increased by all the extra
mitochondria. Enzymes work better at higher temperatures, but icefish enzymes are presumably adapted to
work at very low temperatures (enzyme studies on icefish are carried out close to zero degrees C), but
activity still seems low compared to enzymes from other fish at 0C. The activity of some enzymes is also
elevated, for example lactate dehydrogenase (an enzyme of anaerobic metabolism) seems particularly
active. Perhaps we are seeing a partial transition from aerobic to anaerobic metabolism in these aerobic
muscle fibres.
So why so many mitochondria? It seems that they must be there for some function other than aerobic
respiration, or else why have more mitochondria that are less efficient than they need be and which increase
muscle fibre diameter, making access to oxygen more difficult? One possible answer is the fact that
mitochondria are lipid-rich - they have a high density of lipid membranes and oxygen is more soluble in lipid.
Most of the mitochondria are present in the outer cytoplasm of the muscle fibre (sarcoplasm) forming a
sheath beneath the cell membrane (sarcolemma) and around the contractile core of protein myofibrils, as
shown in model (B) above. Might this enhance oxygen absorption? One caveat - mitochondria may have
membranes consisting of only 20% lipid and 80% protein and so this needs to be considered as lipid is more
permeable to oxygen than water, but protein less so. However, as we have seen, the enzyme (and hence
presumably the total protein content) of the mitochondrial membranes is greatly reduced which supports the
idea that the mitochondria are indeed transporting oxygen into the muscle fibre.
A more complex mathematical model has been developed which incorporates the factors we have discussed:
protein content, lipid content, the outer sheath of mitochondria, myoglobin content, permeability to oxygen,
and a whole manner of other factors. This model relates oxygen concentration to permeability to oxygen
coming from the capillaries (sources) and oxygen consumption by mitochondria (sinks). Oxygen consumption
clearly lowers oxygen content but also increases the diffusion gradient which may accelerate oxygen
diffusion. This model attempts to balance these factors and create an estimate of intracellular oxygen
concentration in the steady-state (that is when supply and consumption are balanced so that oxygen
concentration does not change with time). Needles to say, the mathematics is rather complicated!
Some results from this model are shown below:
This crude model illustrates some key basics. At higher temperatures, for example, in which oxygen diffuses
faster, we might expect more oxygen to reach the middle of the muscle fibre. However, in reality this is slightly
more than compensated for by the increased solubility of oxygen in cold water - the permeability of oxygen
(diffusion coefficient x solubility) actually increases slightly in the cold. Permeability replaces the diffusion
coefficient in our model. Most vertebrate muscle also contains myoglobin, a pink oxygen-binding pigment
which acts as a store of oxygen and also increases transport of oxygen across the muscle fibre. However,
perhaps paradoxically icefish muscles usually have no myoglobin. Chaenocephalus aceratus has traces of
myoglobin in its heart muscle, but none at all in its swimming muscles! Oxygen is more soluble in lipid
(oil) than in water. Muscle fibres are sometimes rich in lipid droplets, which as well as being a store of fuel
may also serve to increase permeability to oxygen, allowing more oxygen to reach the muscle fibre centre.
However, icefish muscle fibres have low lipid content! However, they also have a reduced protein content.
protein significantly impedes oxygen transport across muscle fibres, and the more watery nature of
icefish muscles is expected to significantly increase permeability to oxygen.
Mitochondria and muscle fibre diameter
Distance between the capillaries, or in other words muscle fibre diameter, is also important. In larger fibres
the oxygen has further to travel and this lowers the oxygen concentration in the middle of the fibre (all other
things being equal). Fish grow by increasing muscle fibre diameter, rather than by adding new fibres, so
older and bigger fish have larger muscle fibres and bigger fish tend to rely increasingly less on aerobic
respiration, becoming more anaerobic. Aerobic red muscle fibres are also much smaller than anaerobic white
fibres, since red fibres need faster oxygen delivery. Paradoxically, the red fibres of icefish are unusually
large, larger than is typical for a red-blooded fish of the same size! However, this increase is not chiefly due
to an increase in contractile muscle proteins, nut in mitochondria. Icefish muscles are packed with
mitochondria which account for about 50% of the muscle fibre volume in aerobic swimming muscles.
This proliferation of mitochondria accounts for the large size of the muscle fibres.
This is naively intuitive - it seems as if icefish muscles have more mitochondria to use more oxygen and
make more ATP. However, it is not so simple! For a start, where does the oxygen that the mitochondria need
come from? Detailed analysis has also shown that the mitochondria of icefish have an usually low cristae
density, reduced by 30% (so total cristae surface area is only slightly higher than usual) and enzyme studies
also show that aerobic respiration enzyme activity per gram of mitochondria is about 50% of normal, so the
maximum possible oxygen consumption by the muscle seems normal and is not increased by all the extra
mitochondria. Enzymes work better at higher temperatures, but icefish enzymes are presumably adapted to
work at very low temperatures (enzyme studies on icefish are carried out close to zero degrees C), but
activity still seems low compared to enzymes from other fish at 0C. The activity of some enzymes is also
elevated, for example lactate dehydrogenase (an enzyme of anaerobic metabolism) seems particularly
active. Perhaps we are seeing a partial transition from aerobic to anaerobic metabolism in these aerobic
muscle fibres.
So why so many mitochondria? It seems that they must be there for some function other than aerobic
respiration, or else why have more mitochondria that are less efficient than they need be and which increase
muscle fibre diameter, making access to oxygen more difficult? One possible answer is the fact that
mitochondria are lipid-rich - they have a high density of lipid membranes and oxygen is more soluble in lipid.
Most of the mitochondria are present in the outer cytoplasm of the muscle fibre (sarcoplasm) forming a
sheath beneath the cell membrane (sarcolemma) and around the contractile core of protein myofibrils, as
shown in model (B) above. Might this enhance oxygen absorption? One caveat - mitochondria may have
membranes consisting of only 20% lipid and 80% protein and so this needs to be considered as lipid is more
permeable to oxygen than water, but protein less so. However, as we have seen, the enzyme (and hence
presumably the total protein content) of the mitochondrial membranes is greatly reduced which supports the
idea that the mitochondria are indeed transporting oxygen into the muscle fibre.
A more complex mathematical model has been developed which incorporates the factors we have discussed:
protein content, lipid content, the outer sheath of mitochondria, myoglobin content, permeability to oxygen,
and a whole manner of other factors. This model relates oxygen concentration to permeability to oxygen
coming from the capillaries (sources) and oxygen consumption by mitochondria (sinks). Oxygen consumption
clearly lowers oxygen content but also increases the diffusion gradient which may accelerate oxygen
diffusion. This model attempts to balance these factors and create an estimate of intracellular oxygen
concentration in the steady-state (that is when supply and consumption are balanced so that oxygen
concentration does not change with time). Needles to say, the mathematics is rather complicated!
Some results from this model are shown below:
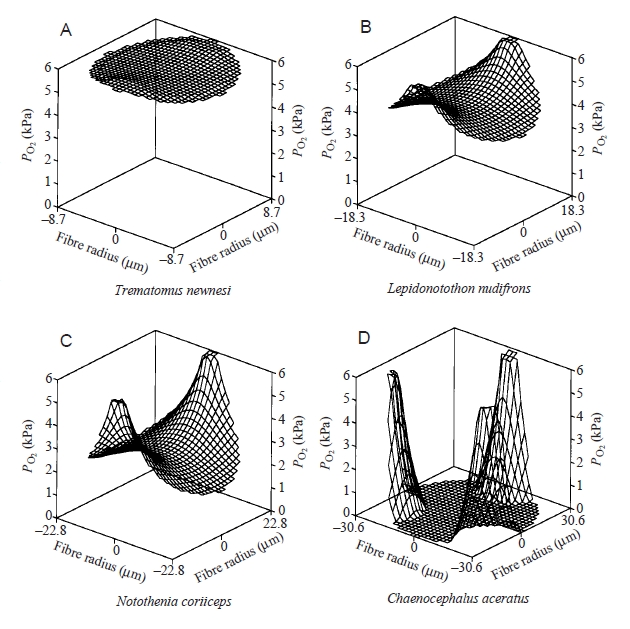
This figure compares oxygen levels, calculated from a mathematical model, in the muscle fibres of
three red-blooded Antarctic fish and one icefish. Fish of a similar size are compared in each case.
In each plot the oxygen concentration (measured as pressure of oxygen or PO2) is calculated
across a single muscle fibre. Trematomus newnesi is a red-blooded Antarctic fish and has one
capillary supplying each fibre, on average; Lepidonotothon nudifrons is also a red-blooded
Antarctic fish with 2 capillaries around each muscle fibre; Notothenia coriiceps is a red-blooded
Antarctic fish with reduced haematocrit and has 2 capillaries around each fibre; and
Chaenocephaluis aceratus 3.
This figure appeared in: Egginton et al., 2002 (copyright: The Company of Biologists limited).
three red-blooded Antarctic fish and one icefish. Fish of a similar size are compared in each case.
In each plot the oxygen concentration (measured as pressure of oxygen or PO2) is calculated
across a single muscle fibre. Trematomus newnesi is a red-blooded Antarctic fish and has one
capillary supplying each fibre, on average; Lepidonotothon nudifrons is also a red-blooded
Antarctic fish with 2 capillaries around each muscle fibre; Notothenia coriiceps is a red-blooded
Antarctic fish with reduced haematocrit and has 2 capillaries around each fibre; and
Chaenocephaluis aceratus 3.
This figure appeared in: Egginton et al., 2002 (copyright: The Company of Biologists limited).
Notice the unusual features of the icefish model prediction: large muscle fibres, 3 capillaries per fibre, and an
expected oxygen pressure rapidly reaching zero inside the muscle fibre! However, the centre of these muscle
fibres have mitochondria and so must have a minimum PO2 of 0.3 kPa. The only factor we have discussed,
which is not included in this model, is the reduction in mitochondria cristae and enzyme function, which raises
the PO2 to just enough for the mitochondria to function.
One final factor that we know of which has not been factored in is illustrated in model (C) above: mitochondria
are also aligned in rays or spokes that radiate from the centre of the muscle fibre, outlining individual
myofibrils within the muscle. If the mitochondria are indeed serving to transport oxygen then this arrangement
is expected to increase oxygen levels across the muscle and to reach central mitochondria. However,
modeling such a system appears difficult!
An evolutionary accident?
Sidell and O'Brien (2006) have argued that the loss of erythrocytes in icefish may be an evolutionary
accident. They base this conclusion on a calculation of energy expenditure of the heart by multiplying cardiac
output by arterial blood pressure. The icefish has slightly lower arterial blood pressure than red-blooded
Antarctic fish, however, in the absence of actual data for venous pressure the actual pressure head
(pressure drop in one circuit of the blood) could not be calculated and was assumed close to zero and similar
to that of red-blooded Antarctic fish. With the large heart and large blood-volume the calculation produces
the result that the icefish heart utilises twice as much energy per cycle as that of red-blooded Antarctic fish.
However, since the capillaries, the narrowest vessels in the circuit,of the icefish are 2-3 times the diameter of
those of red-blooded fish one would expect the pressure drop across them to be much smaller. According to
Poiseuille's equation for flow in a smooth-walled cylindrical pipe:
expected oxygen pressure rapidly reaching zero inside the muscle fibre! However, the centre of these muscle
fibres have mitochondria and so must have a minimum PO2 of 0.3 kPa. The only factor we have discussed,
which is not included in this model, is the reduction in mitochondria cristae and enzyme function, which raises
the PO2 to just enough for the mitochondria to function.
One final factor that we know of which has not been factored in is illustrated in model (C) above: mitochondria
are also aligned in rays or spokes that radiate from the centre of the muscle fibre, outlining individual
myofibrils within the muscle. If the mitochondria are indeed serving to transport oxygen then this arrangement
is expected to increase oxygen levels across the muscle and to reach central mitochondria. However,
modeling such a system appears difficult!
An evolutionary accident?
Sidell and O'Brien (2006) have argued that the loss of erythrocytes in icefish may be an evolutionary
accident. They base this conclusion on a calculation of energy expenditure of the heart by multiplying cardiac
output by arterial blood pressure. The icefish has slightly lower arterial blood pressure than red-blooded
Antarctic fish, however, in the absence of actual data for venous pressure the actual pressure head
(pressure drop in one circuit of the blood) could not be calculated and was assumed close to zero and similar
to that of red-blooded Antarctic fish. With the large heart and large blood-volume the calculation produces
the result that the icefish heart utilises twice as much energy per cycle as that of red-blooded Antarctic fish.
However, since the capillaries, the narrowest vessels in the circuit,of the icefish are 2-3 times the diameter of
those of red-blooded fish one would expect the pressure drop across them to be much smaller. According to
Poiseuille's equation for flow in a smooth-walled cylindrical pipe:
Doubling the radius is expected to cause a (2^4) 16-fold reduction in the pressure drop across the
capillaries. The capillaries may be slightly longer due to increased turtuosity which would partially counter
this reduction in pressure head, but on the other hand the loss of erythrocytes would be expected to further
reduce the pressure drop since Poiseuille's equation ignores the non-Newtonian effects of red blood cells
squeezing through capillaries which should cause a greater pressure drop. All in all, we might expect the
pressure-drop across the icefish capillary to be 10-15 times less than that across the capillary of a red
blooded fish.
It should be noted that red-blooded Antarctic fish have lower erythrocyte densities than temperate fish, so
there is clearly some selection pressure on these fish to reduce red cell content. It has also been noted that
Antarctic fish do not simply reduce red cell number, but that their red cells tend to have lower haemoglobin
concentrations. This could indicate that the pressure is on haemoglobin reduction for reasons other than to
reduce blood viscosity, however, it could also be a way of making the red blood cells more pliable so that
they pass through the capillaries more easily. Certainly, once icefish lost the majority of their erythrocytes
(and those remaining carry no haemoglobin) then they would no longer need narrow capillaries, since these
function to slow the passage of erythrocytes for gas exchange.
If measurements of venous pressure confirm that the pressure drop during one circuit of the blood is indeed
much lower in the icefish then the energy consumption by the heart may not be as great as imagined and
may even be less than in red-blooded fish. On the other hand, if venous pressure falls greatly then this
requires physical explanation and would indeed suggest the loss of haemoglobin for reasons other than to
reduce blood viscosity. This could be an accidental mutation causing loss of haemoglobin, though it is hard
to see why this would be favoured by selection if it does have huge energetic costs when icefish compete
with red-blooded Antarctic fish. Perhaps there is some other reason for reduction and loss of haemoglobin
synthesis in Antarctic fish, perhaps some problem incurred by the necessary enzymes having to operate at
such low temperatures.
Conclusion
It would appear, from our models, that the icefish has a wide range of adaptations which place its muscles
on the edge of the aerobic limit - they seem not to be supplied with plentiful oxygen, but just enough to
function. However, a model is only as good as its assumptions and data inputs. The physiology of icefish is
extreme, complex and difficult to understand and it is not easy to observe what really happens inside a
living, working muscle fibre. (It would be nice to have actual oxygen concentration measurements from
different regions inside the muscle fibres). However, the model has provided us with many answers and it
seems that a whole suite of adaptations keep icefish functioning well despite their colourless blood.
It should also be noted that icefish do have a regulated reserve aerobic capacity; they are not simply
operating on the edge of aerobic ability all the time. They can increase oxygen consumption by increasing
cardiac output by 50% over resting levels if needed, and are thought to absorb more oxygen by shunting
more blood to the gills.
As a final note, I hope you can see how important it is to study such organisms that are of little economic
importance (some icefish are commercially fished, however). The human race is in real danger of collapsing
into ignorance as the increasing demands of financial constraints is tending to draw more and more funding
from basic science towards commercial only enterprises. Many species in the oceans are yet to be
discovered, and some are going extinct and will likely become extinct before they are known to science.
What a waste to lose all that valuable information about how the greatest engineer of them all, Mother
Nature, solves the challenges of life! Please support basic science, even when it has no immediate
foreseeable commercial value. Who can say what future value knowledge may possess? What kind of
intelligent species lives in ignorance of its own world?
Bibliography/References
Eastman, J.T., 1993. Antarctic fish Biology: Evolution in a unique environment. Pub: Academic press, Inc.
Prisco, G.d., B. Maresca and B. Tota (eds.), 1991. Biology of Antarctic Fish. Pub: Springer-Verlag.
Ruud, T. The Ice Fish.
Hoofd, L. and S. Egginton, 1997. The possible role of intracellular lipid in determining oxygen delivery to fish
skeletal muscle. Respiration Physiol. 107: 191-202.
Egginton, S., C. Skilbeck, L. hoofd, J. Calvo and I.A. Johnston, 2002. Peripheral oxygen transport in skeletal
muscle of Antarctic and sub-Antarctic notothenioid fish. J. Exp. Biol. 205: 769-779.
O'Brien, K.M., C. Skilbeck, B.D. Sidell and S. Egginton, 2003. J. Exp. Biol. 206: 411-421.
Egginton, S., 2000. The influence of environmental temperature on microvascular development in fish.
Zoology:102:164-172.
Hemminsen, E.A. Respiratory and cardiovascular adaptations in haemoglobin-free fish: resolved and
unresolved problems.
Sidell, BD, and K.M. O’Brien, 2006. When bad things happen to good fish: the loss of hemoglobin and
myoglobin expression in Antarctic icefishes. Journal of Experimental Biology 209: 1791-1802.
Article updated: 5/4/2015
capillaries. The capillaries may be slightly longer due to increased turtuosity which would partially counter
this reduction in pressure head, but on the other hand the loss of erythrocytes would be expected to further
reduce the pressure drop since Poiseuille's equation ignores the non-Newtonian effects of red blood cells
squeezing through capillaries which should cause a greater pressure drop. All in all, we might expect the
pressure-drop across the icefish capillary to be 10-15 times less than that across the capillary of a red
blooded fish.
It should be noted that red-blooded Antarctic fish have lower erythrocyte densities than temperate fish, so
there is clearly some selection pressure on these fish to reduce red cell content. It has also been noted that
Antarctic fish do not simply reduce red cell number, but that their red cells tend to have lower haemoglobin
concentrations. This could indicate that the pressure is on haemoglobin reduction for reasons other than to
reduce blood viscosity, however, it could also be a way of making the red blood cells more pliable so that
they pass through the capillaries more easily. Certainly, once icefish lost the majority of their erythrocytes
(and those remaining carry no haemoglobin) then they would no longer need narrow capillaries, since these
function to slow the passage of erythrocytes for gas exchange.
If measurements of venous pressure confirm that the pressure drop during one circuit of the blood is indeed
much lower in the icefish then the energy consumption by the heart may not be as great as imagined and
may even be less than in red-blooded fish. On the other hand, if venous pressure falls greatly then this
requires physical explanation and would indeed suggest the loss of haemoglobin for reasons other than to
reduce blood viscosity. This could be an accidental mutation causing loss of haemoglobin, though it is hard
to see why this would be favoured by selection if it does have huge energetic costs when icefish compete
with red-blooded Antarctic fish. Perhaps there is some other reason for reduction and loss of haemoglobin
synthesis in Antarctic fish, perhaps some problem incurred by the necessary enzymes having to operate at
such low temperatures.
Conclusion
It would appear, from our models, that the icefish has a wide range of adaptations which place its muscles
on the edge of the aerobic limit - they seem not to be supplied with plentiful oxygen, but just enough to
function. However, a model is only as good as its assumptions and data inputs. The physiology of icefish is
extreme, complex and difficult to understand and it is not easy to observe what really happens inside a
living, working muscle fibre. (It would be nice to have actual oxygen concentration measurements from
different regions inside the muscle fibres). However, the model has provided us with many answers and it
seems that a whole suite of adaptations keep icefish functioning well despite their colourless blood.
It should also be noted that icefish do have a regulated reserve aerobic capacity; they are not simply
operating on the edge of aerobic ability all the time. They can increase oxygen consumption by increasing
cardiac output by 50% over resting levels if needed, and are thought to absorb more oxygen by shunting
more blood to the gills.
As a final note, I hope you can see how important it is to study such organisms that are of little economic
importance (some icefish are commercially fished, however). The human race is in real danger of collapsing
into ignorance as the increasing demands of financial constraints is tending to draw more and more funding
from basic science towards commercial only enterprises. Many species in the oceans are yet to be
discovered, and some are going extinct and will likely become extinct before they are known to science.
What a waste to lose all that valuable information about how the greatest engineer of them all, Mother
Nature, solves the challenges of life! Please support basic science, even when it has no immediate
foreseeable commercial value. Who can say what future value knowledge may possess? What kind of
intelligent species lives in ignorance of its own world?
Bibliography/References
Eastman, J.T., 1993. Antarctic fish Biology: Evolution in a unique environment. Pub: Academic press, Inc.
Prisco, G.d., B. Maresca and B. Tota (eds.), 1991. Biology of Antarctic Fish. Pub: Springer-Verlag.
Ruud, T. The Ice Fish.
Hoofd, L. and S. Egginton, 1997. The possible role of intracellular lipid in determining oxygen delivery to fish
skeletal muscle. Respiration Physiol. 107: 191-202.
Egginton, S., C. Skilbeck, L. hoofd, J. Calvo and I.A. Johnston, 2002. Peripheral oxygen transport in skeletal
muscle of Antarctic and sub-Antarctic notothenioid fish. J. Exp. Biol. 205: 769-779.
O'Brien, K.M., C. Skilbeck, B.D. Sidell and S. Egginton, 2003. J. Exp. Biol. 206: 411-421.
Egginton, S., 2000. The influence of environmental temperature on microvascular development in fish.
Zoology:102:164-172.
Hemminsen, E.A. Respiratory and cardiovascular adaptations in haemoglobin-free fish: resolved and
unresolved problems.
Sidell, BD, and K.M. O’Brien, 2006. When bad things happen to good fish: the loss of hemoglobin and
myoglobin expression in Antarctic icefishes. Journal of Experimental Biology 209: 1791-1802.
Article updated: 5/4/2015



